As France enters near-total lockdown on Tuesday, researchers worldwide are racing to develop a COVID-19 vaccine. This Monday, a U.S. team initiated the first clinical trial for a groundbreaking candidate.
Traditional vaccine development often uses weakened or inactivated viruses to train the immune system. The goal is to expose the body to viral proteins, enabling it to recognize and fight the pathogen in future encounters—without causing illness.
However, isolating, modifying, and scaling up virus production is time-intensive and complex.
For viruses with few surface proteins that trigger immunity, scientists can instead produce those proteins directly using recombinant DNA technology. While faster, large-scale manufacturing remains challenging.
A more innovative method, available for years, accelerates the process dramatically: it enables vaccine design immediately after sequencing the virus's genome—as done by the Institut Pasteur in Wuhan weeks ago.
Proteins are synthesized by translating messenger RNA (mRNA), transcribed from genetic material. Viruses hijack our cells by delivering their mRNA blueprint to produce viral proteins.
This mRNA-based approach copies the virus's key mRNAs, packages them into a vaccine, and injects them. Once inside cells, they prompt production of harmless viral proteins, priming the immune system for real infection.
These proteins can't replicate or harm without the full virus. The immune system "remembers" them, mounting a swift defense later.
Challenges include the immune system's tendency to destroy foreign RNA and RNA's difficulty crossing cell membranes. Solutions: produce excess mRNA and encapsulate it in lipid nanoparticles that fuse with cells for efficient delivery.
Biotech leader Moderna mastered these hurdles with mRNA-1273, an experimental COVID-19 vaccine. Phase I trials, led by the Kaiser Permanente Washington Research Institute in Seattle, began testing safe doses in 45 healthy adults aged 18-55.
The first volunteers: Neal Browning, a man, and Jennifer Haller, COO of a tech firm. "We all feel so helpless. This is my chance to contribute," she shared.
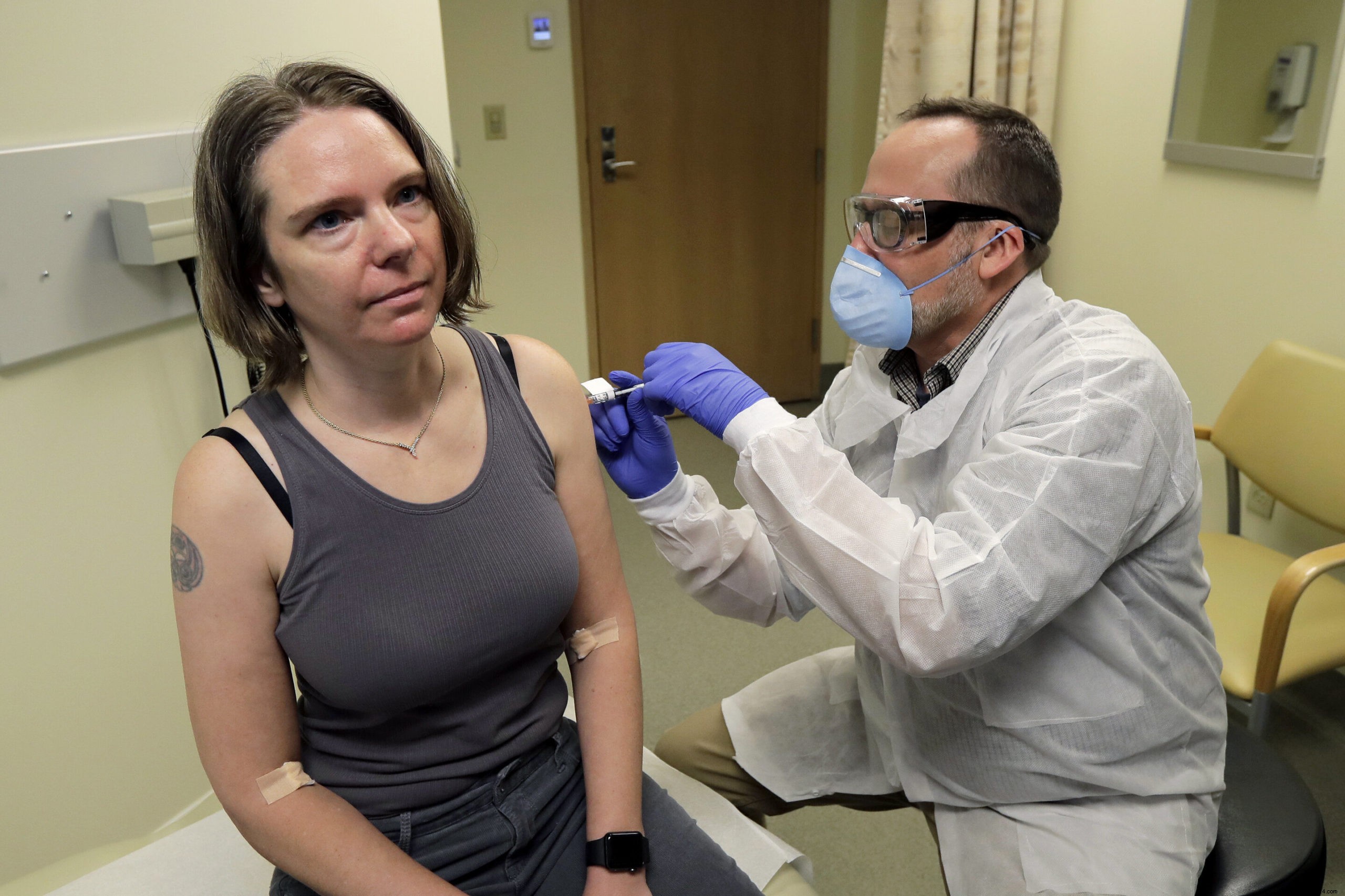
If Phase I succeeds, Phase II will evaluate immune responses in more participants. Moderna is scaling production. Success could yield a widely available vaccine in 12-18 months.
Source
Related Articles: