Lab-grown mini-organs hold immense promise for unraveling disease mechanisms and testing novel therapies. Researchers at Cincinnati Children's Hospital Medical Center have achieved a milestone with the most sophisticated gastric organoids yet, detailed in Cell Stem Cell.
While cell cultures and animal models have advanced medicine, they often fall short in predicting human outcomes. Enter organoids: tiny, 3D organs derived from stem cells that offer a more accurate human-relevant model.
Recent years have seen remarkable strides. Two years ago, scientists grew genetically modified mini-livers to study non-alcoholic fatty liver disease, marked by excess liver fat.
Now, the Cincinnati team has pioneered advanced gastric organoids, pushing the boundaries of this emerging field.
Starting with human pluripotent stem cells—which can be reprogrammed to an embryonic-like state and directed into any cell type—the researchers differentiated them into three key precursors for stomach development: neuroglial, mesenchymal, and epithelial enteric cells.
"From these, we generated gastric tissue containing acid-producing glands, surrounded by layers of smooth muscle with functional enteric neurons that controlled contractions in modified antral gastric tissue," explains lead author Alexandra Eicher.
After roughly 30 days in culture, the organoids were transplanted into mice. Benefiting from blood supply and ample space, they expanded over 1,000 times larger than in vitro.
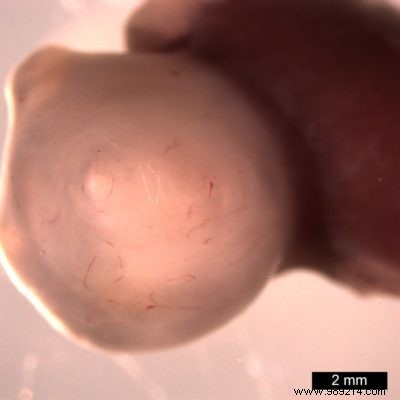
Remarkably, these mini-organs developed features like Brunner's glands, which secrete protective alkaline mucus to shield the upper intestine from stomach acid—a trait impossible in standard cultures.
The multi-precursor approach could extend to other organoids, with long-term potential for growing full-sized, functional organs. This could transform transplantation, disease modeling, and drug testing.