Researchers have uncovered how certain bacteria adapt to prolonged antibiotic exposure by dramatically altering their shape. This groundbreaking study appears in Nature Physics.
For decades, antibiotics have saved millions of lives by combating bacterial infections. Yet the battle is far from over. Bacteria continuously evolve, developing resistance through overuse of these vital drugs.
As a result, treatments are losing effectiveness. Scientists are raising alarms, urging stricter controls on antibiotic use to prevent a total breakdown. The World Health Organization warns that resistant bacteria could claim up to 10 million lives annually by 2050.
New strategies are urgently needed to thwart these infections. Experts are dissecting bacterial defense mechanisms, including well-known lateral gene transfer—the primary driver of resistance spread. A team led by Shiladitya Banerjee at Carnegie Mellon University has now identified a novel adaptation.
The study examined how antibiotics impact the growth and form of Caulobacter crescentus, a key model bacterium. Exposure to non-lethal doses of chloramphenicol—a broad-spectrum antibiotic—over multiple generations allowed these bacteria to "recover their pre-stimulus growth rate while undergoing dramatic changes in cell shape," the authors report.
In essence, after about 10 generations under low-dose pressure, the bacteria curled inward, radically reshaping themselves. Once antibiotics were removed, cells reverted to their original form over several generations.
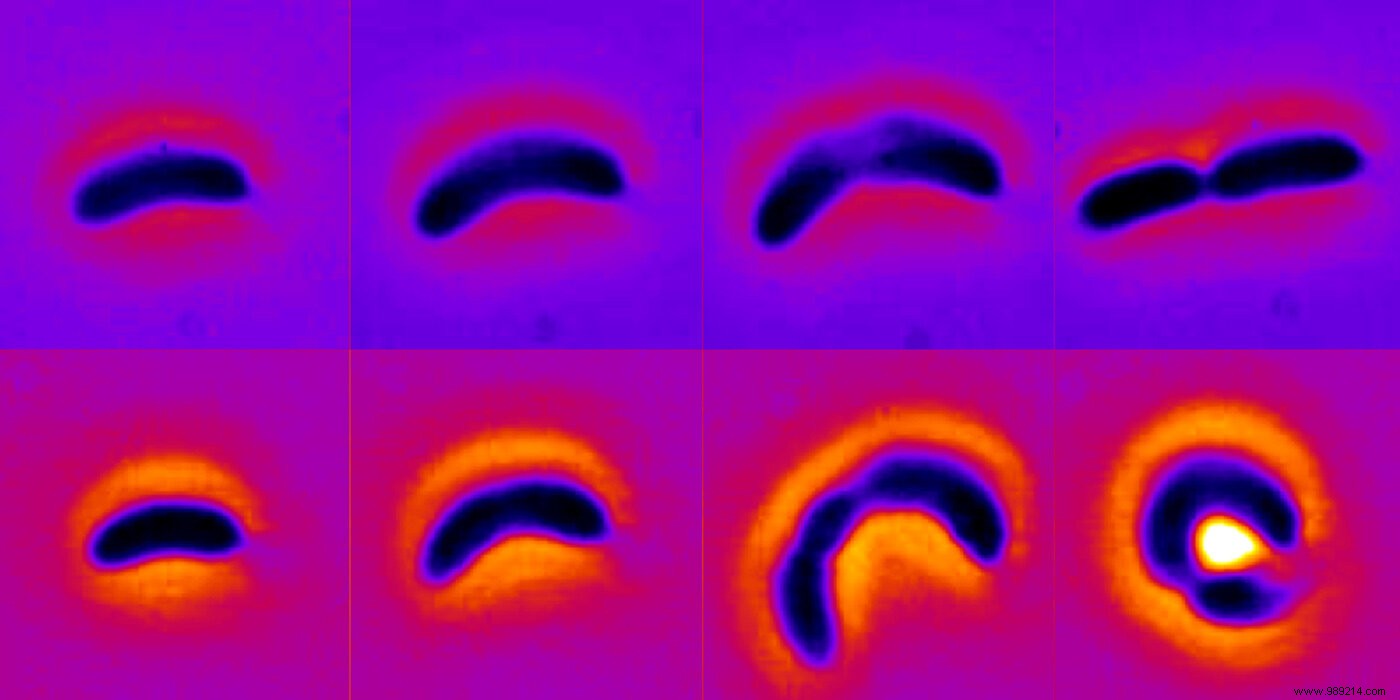
Single-cell experiments and theoretical modeling reveal that increased cell width boosts volume, diluting internal antibiotics. Curvature and width also reduce the surface-area-to-volume ratio, limiting antibiotic entry through the cell membrane.
"This research holds major implications for human health and will likely spur further molecular studies on how cell shape influences bacterial growth and antibiotic resistance," Banerjee concludes.