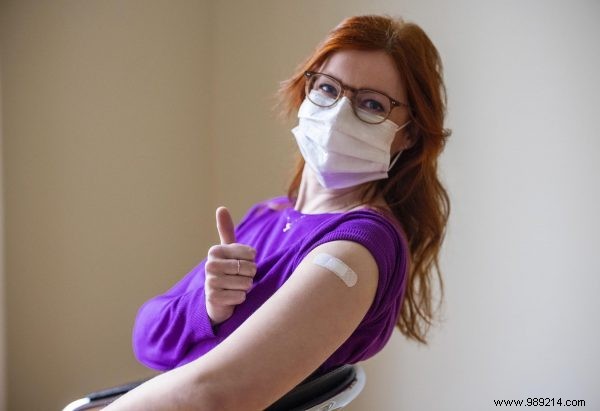
We're temporarily pausing AstraZeneca COVID-19 vaccinations for a few weeks as a precaution while investigating reported side effects. With multiple vaccines now available in Europe, do you have a preference?
The AstraZeneca vaccine has been suspended amid reports of potential blood clots in several countries. The Ministry of Health explains: "The immediate reason for the advice is new information that became available this weekend. It concerns complaints other than the limited number of reports of thrombosis after vaccination, which the MEB based on this weekend."
The Dutch Medicines Evaluation Board (MEB) notes no proven link between the vaccine and blood clot cases reported in Norway and Denmark. They urge anyone vaccinated with AstraZeneca to contact their GP promptly if unusual symptoms appear after three days, such as unexplained complaints or blue spots on the skin.
Outgoing Health Minister Hugo de Jonge confirms the vaccine's overall safety, emphasizing this as a precautionary step.
Europe has authorized several effective vaccines. Here's a clear overview based on official EMA data:
The first approved in Europe, this mRNA vaccine trains body cells to produce a harmless coronavirus protein. This prepares your immune system to recognize and neutralize the virus, delivering over 90% protection.
The second mRNA vaccine authorized, it works similarly to Pfizer/BioNTech and also provides more than 90% protection.
This viral vector vaccine uses a modified chimpanzee cold virus to trigger production of coronavirus spike proteins, activating immunity. The EMA reports 60% protection.
This Dutch-developed viral vector vaccine, recently EMA-approved, requires just one dose and mirrors AstraZeneca's mechanism, offering 66% protection.
Vaccines are typically allocated by age, health conditions, or priority groups like healthcare workers.
If given the choice, which vaccine would you prefer?